On 15th December the Board of Member States of European Reference Networks (ERNs) voted to approve all 23 ERN proposals submitted under the first wave (the 24th proposal, submitted under the 2nd call, will be assessed early in 2017).
This momentous step comes after years of collaboration and efforts between rare disease patients, clinical experts and policy makers in EU Member States, at the European Commission and the European Parliament to bring the ERNs to fruition.
European Reference Networks are networks connecting expert centres in the field of rare diseases and specialised healthcare, organised across borders. This concept has been developing and maturing over the past five years and represents a major innovation in care for Europe’s 30 million rare disease patients: although pan-European structures exist in the research domain, this is the first such enterprise in the health sphere. The Networks will create governance structures for knowledge sharing and care coordination across the EU to improve access to diagnosis and treatment, as well as high-quality healthcare provision. ERNs have been organised around broad disease groups, to ensure that no patient with a rare disease is left ‘without a home’ under an ERN.
This is a major innovation for rare diseases in Europe, involving 370 hospitals and almost 1000 highly specialised units across 26 Countries (25 EU Member States + Norway).
The full list of approved ERN applications is available via the official European Commission webpage.
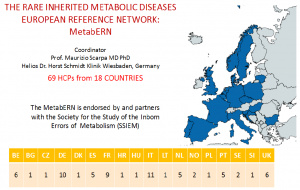
The European Reference Network for Hereditary Metabolic Diseases (MetabERN) represents the first most comprehensive, pan-metabolic, pan-european, patient-orientated platform ever conceived, that will transform how care is provided to patients with inherited metabolic diseases (IMDs) in Europe.
- MetabERN represents 69 founding nationally certified Healthcare Providers from 18 European countries, 44 Patients Organisations and is endorsed by the Society for the Inborn Errors of Metabolism.
- MetabERN involves 1681 experts, of which about 52% are specialized medical doctors (particularly pediatrician, geneticist, neurologist and metabolic physicians).
- 42.427 are the patients managed by the MetabERN, 68%of which represented by pediatric patients. More than a half of the patients are affected by lysosomal disorders or aminoacid and organic acids related disorders diseases (23% and 39% respectively).
All Inherited Metabolic Diseases (IMDs), with no exclusion, are of interest for the MetabERN, independently from their prevalence, frequency and existing previous interest for research or therapy development.
Considering the complexity of the IMDs field as a whole, the group of more than 700 ultra rare metabolic diseases are structured in 7 subnetworks:
1. Aminoacid and organic acids related disorders
2. Disorders of pyruvate metabolism, Krebs cycle defects, mitochondrial oxidative phosphorylation disorders, disorders of thiamine transport and metabolism
3. Carbohydrate fatty acid oxidation and ketone bodies disorders
4. Lysosomal disorders
5. Peroxisomal and lipid related disorders
6. Congenital disorders of glycosylation and disorders of intracellular trafficking
7. Disorders of Neuromodulators and Small Molecules.
In disease-specific networks professionals and patient associations will collaborate to transversal programs common to all the subnetworks. Patient organizations will work in close collaboration with Healthcare Professionals to map and understand patients’ needs and identify solutions.
MetabERN aims to:
- Accommodate and interconnect expertise across Europe at the service of IMD patients,
- Harmonise data collection across participating European centres,
- Establish common approaches to optimise prevention, diagnostics, management and treatment,
- Develop and implement harmonized guidelines,
- Stimulate cross-border research and innovative treatments
- Develop training and education opportunities especially in Member States where it is lacking,
- Interact closely with patients to serve as a reliable source of information and involve them in the planning of their individual care pathways and decision-making.
For these reasons, the MetabERN will be instrumental to facilitate and harmonize patients’ access to diagnosis and best treatment and to designing a collaborative govermental structure with patients, academia, politics, insurance companies and industry.
For more information please contact
Prof. Maurizio Scarpa, MD, PhD
MetabERN Coordinator
Maurizio.Scarpa@helios-kliniken.de